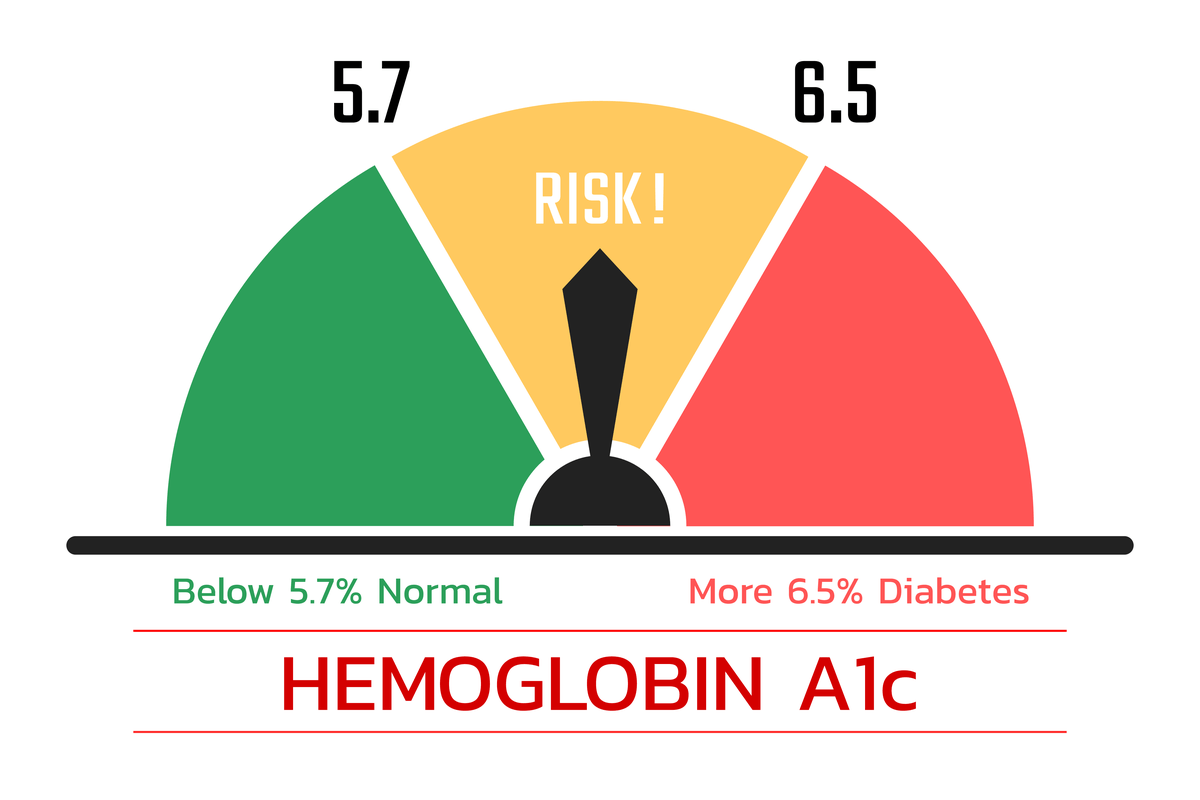
Last updated on September 1st, 2022Poor lifestyle and eating habits can lead to the risk of developing diabetes or pre-diabetes. If not taken care of, diabetes can lead to several health complications like cardiovascular disease, kidney damage, stress, depression and makes your life worse. Therefore, you should keep a check on your blood sugar levels. On …
Last updated on April 12th, 2024The amount of glucose (“sugar” expressed in mg/dL) present in the blood fluctuates during the day and at night. Our body maintains a level of blood glucose for metabolism. According to the American Diabetes Association, ,the normal sugar level in a healthy body is less than 100 mg/dL. But sometimes, these …
Last updated on November 3rd, 2023What Is HbA1c? HbA1c full form is hemoglobin A1c or glycosylated hemoglobin. It is a form of hemoglobin that contains sugar. Hemoglobin is a kind of molecule that is present in red blood cells and helps in carrying oxygen to the body’s tissue. Read this blog to know what the hba1c …
Blood sugar, also known as blood glucose, is the fuel that keeps our bodies running. It’s a simple sugar derived from the carbohydrates we consume, and it plays a central role in energy production at the cellular level. Maintaining healthy blood sugar levels is essential for overall well-being. However, when blood sugar levels rise above a …
The hs-CRP test, also known as the high-sensitivity C-reactive protein test, is a powerful diagnostic tool that can provide valuable insights into your overall health. This simple blood test gauges the levels of C-reactive protein, a biomarker that indicates inflammation within the body. By understanding your hs-CRP levels, you and your doctor can gain important clues …
The human body is a marvel of complex organs working together to function properly. One of the essential organs of the human body is its kidneys. These bean-shaped organs quietly perform vital functions, filtering waste and excess fluid from the blood. However, like any well-oiled machine, the kidneys can falter, leading to potentially serious consequences. The …
Welcome to our in-depth exploration of the insulin fasting test – a crucial diagnostic test in understanding metabolic health and assessing insulin resistance. In today’s world, where lifestyle factors and dietary habits play significant roles in health outcomes, comprehending insulin’s role in the body is paramount. In this blog, we delve into the complications of insulin …
Your kidneys are responsible for many important bodily functions. They filter out waste products, excess fluids, and toxins from your blood. They play a vital role in regulating electrolyte levels, such as sodium, potassium, and phosphorus, and maintaining a healthy balance of these minerals is crucial for kidney function. Chronic Kidney Disease (CKD) is a condition …


 English
English